 
In accord with this, enrichment of the siRNA effector Argonaute2 (Ago2) was found at some 1.688(X) repeats. This study postulatex that siRNA-dependent modification of 1.688(X) chromatin contributes to recognition of nearby genes. Interestingly, the siRNA pathway and siRNA from a 1.688(X) repeat also promote X recognition. The 1.688(X) repeats and CES are dissimilar, and appear to operate through different mechanisms. 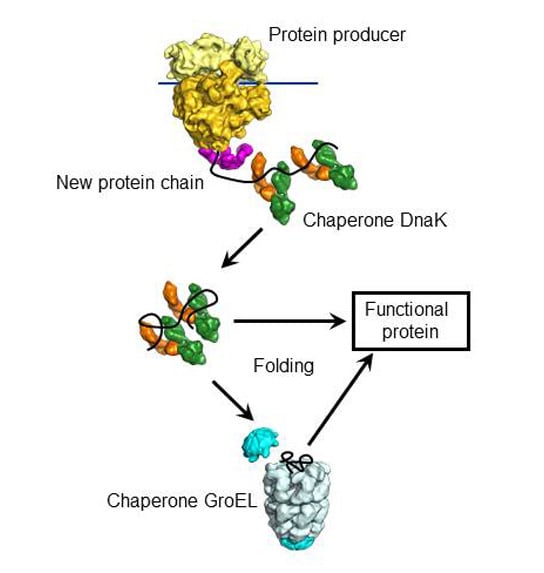
In addition, a family of satellite repeats that is enriched on the X chromosome, the 1.688(X) repeats, promotes recruitment of the complex to nearby genes. The Chromatin Entry Sites (CES) recruit a chromatin-modifying complex that spreads into nearby genes and increases expression. Identification of this chromosome involves cooperation between different classes of X-identity elements. 
Drosophila melanogaster males increase expression from their single X chromosome. How these factors are localized to a chromosome with requisite accuracy is poorly understood. This requires selective recruitment of regulatory factors to the modulated chromosome. Many heterogametic organisms adjust sex chromosome expression to accommodate differences in gene dosage. Chromatin that guides dosage compensation is modulated by the siRNA pathway in Drosophila melanogaster. These data uncover distinct and coordinated actions of the chaperone machinery, where the Hsp70 system expands the structural ensembles of Ago2 and the Hsp90 system captures and stabilizes the active form.ĭeshpande, N. Instead, the Hsp90 system is required to extend the dwell time of Ago2 in the open state, which must be transiently primed by the Hsp70 system. The Hsp70 system, but not the Hsp90 system alone, is sufficient for Ago2 to partially populate the open form. The Hsp70 system ( Hsp40 and Hsp70) and the Hsp90 system ( Hop, Hsp90, and p23) together render Ago2 into an open, active form. Empty Ago2 exists in various closed conformations. This study utilized single-molecule Forster resonance energy transfer (smFRET) to probe the conformational changes of Drosophila Ago2 mediated by the chaperone machinery. This machinery also activates many other clients, including steroid hormone receptors and kinases, but how their structures change during chaperone-dependent activation remains unclear. Loading of small RNAs into argonaute, the core protein in RNA silencing, requires the Hsp70/Hsp90 chaperone machinery. Conformational activation of Argonaute by distinct yet coordinated actions of the Hsp70 and Hsp90 chaperone systems. Keywords - RNA interference, RISC complex, RISC activity, endonuclease, regulation of chromatin insulatorsĬlassification - PIWI domain (endonuclease V protein family) and PAZ domain 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
